Tin - 50Sn: the essentials
- Name: tin
- Symbol: Sn
- Atomic number: 50
- Relative atomic mass (Ar): 118.710 (7) g [see note g]
- Standard state: solid at 298 K
- Appearance: silvery lustrous grey
- Classification: Metallic
- Group in periodic table: 14
- Group name: (none)
- Period in periodic table: 5
- Block in periodic table: p
- Shell structure: 2.8.18.18.4
- CAS Registry: 7440-31-5
Tin atoms have 50 electrons and the shell structure is 2.8.18.18.4. The ground state electronic configuration of neutral tin is [Kr].4d10.5s2.5p2 and the term symbol of tin is 3P0.
Tin: description
Ordinary tin is a silvery-white metal, is malleable, somewhat ductile, and has a highly crystalline structure. Due to the breaking of these crystals, a "tin cry" is heard when a bar is bent. The element has two allotropic forms. On warming, grey, or α-tin, with a cubic structure, changes at 13.2°C into white, or β-tin, the ordinary form of the metal. White tin has a tetragonal structure. When tin is cooled below 13.2°C, it changes slowly from white to grey. This change is affected by impurities such as aluminium and zinc, and can be prevented by small additions of antimony or bismuth. The conversion was first noted as growths on organ pipes in European cathedrals, where it was thought to be the devils work. This conversion was also speculated to be caused microorganisms and was called "tin plague" or "tin disease".
Tin resists distilled, sea, and soft tap water, but is attacked by strong acids, alkalis, and acid salts. Oxygen in solution accelerates the attack. When heated in air, tin forms SnO2. It is, or was, used to plate steel, making "tin cans". Tin is used as one component in bell metals.

Tin: physical properties
Density of solid: 7310 kg m-3
Molar volume: 16.29 cm3
Thermal conductivity: 66.6 (polycrystalline; 515 parallel to c axis, 745 perpendicular to c axis) W m‑1 K‑1
Tin: heat properties
Melting point: 505.08 [231.93 °C (449.47 °F)] K
Boiling point: 2875 [2602 °C (4716 °F)] K
Enthalpy of fusion: 20.5 kJ mol-1
Tin: atom sizes
Atomic radius (empirical): 145 pm
Molecular single bond covalent radius: 140 (coordination number 4) ppm
van der Waals radius: 242 ppm
Tin: electronegativities
Pauling electronegativity: 1.96 (Pauling units)
Allred Rochow electronegativity: 1.72 (Pauling units)
Mulliken-Jaffe electronegativity: 2.21 (sp3 orbital)
Tin: orbital properties
First ionisation energy: 708.58 kJ mol‑1
Second ionisation energy: 1411.88 kJ mol‑1
Third ionisation energy: 2943.4 kJ mol‑1
Tin: abundances
Universe: 4 ppb by weight
Crustal rocks: 2200 ppb by weight
Human: 200 ppb by weight
Tin: crystal structure

Tin: biological data
Human abundance by weight: 200 ppb by weight
Tin might be a necessary element in very, very, small quantities in rats. Organotin compounds are used as bactericides and fungicides in marine environments but cause environmental concern as they cause severe problems to local wildlife.
Tin: uses
Tin: reactions
Reactions of tin as the element with air, water, halogens, acids, and bases where known.
Tin: binary compounds
Binary compounds with halogens (known as halides), oxygen (known as oxides), hydrogen (known as hydrides), and other compounds of tin where known.
Tin: compound properties
Bond strengths; lattice energies of tin halides, hydrides, oxides (where known); and reduction potentials where known.
Tin: history
Tin was discovered by Known since ancient times in unknown at not known. Origin of name: from the Anglo-Saxon word "tin" (the origin of the symbol Sn comes from the Latin word "stannum" meaning "tin").Tin: isotopes
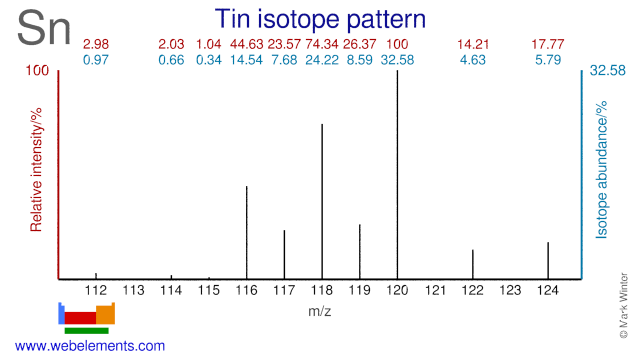
Tin has the most stable isotopes (10) of all elements. Tin isotopes are used in a variety of applications. Sn-112 is used as precursor in the production of the radioisotope Sn-113 while Sn124 is used for producing Sb-124. Sn-116 and Sn-117 can both be used for the production of the medical radioisotope Sn-117m which is used in treating bone cancer. Both Sn-118 and Sn-119 have been evaluated for the production of Sn-119m.
Tin: isolation
Isolation: tHere is normally little need to isolate tin metal in the laboratory as it is readily available commercially. Tin is commonly available as the mineral cassiterite, SnO2. Reduction of this dioxide with burning coal results in tin metal and was probably how tin was made by the ancients.
SnO2 + 2C → Sn + 2CO
