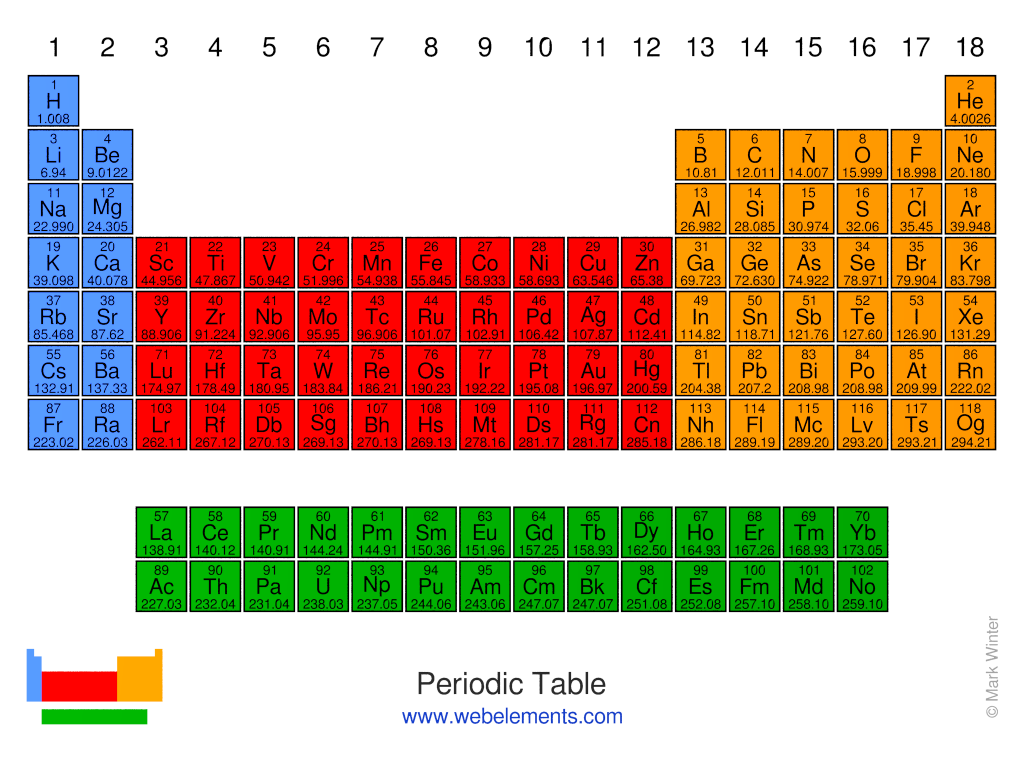
The periodic table of the elements
The periodic table is an arrangment of the chemical elements ordered by atomic number so that periodic properties of the elements (chemical periodicity) are made clear.
Explore the chemical elements through this periodic table
| Group | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Period 1 |
1
1.008
Hydrogen
|
2
4.0026
Helium
|
|||||||||||||||||
| 2 |
3
6.94
Lithium
|
4
9.0122
Beryllium
|
5
10.81
Boron
|
6
12.011
Carbon
|
7
14.007
Nitrogen
|
8
15.999
Oxygen
|
9
18.998
Fluorine
|
10
20.180
Neon
|
|||||||||||
| 3 |
11
22.990
Sodium
|
12
24.305
Magnesium
|
13
26.982
Aluminium
|
14
28.085
Silicon
|
15
30.974
Phosphorus
|
16
32.06
Sulfur
|
17
35.45
Chlorine
|
18
39.948
Argon
|
|||||||||||
| 4 |
19
39.098
Potassium
|
20
40.078
Calcium
|
21
44.956
Scandium
|
22
47.867
Titanium
|
23
50.942
Vanadium
|
24
51.996
Chromium
|
25
54.938
Manganese
|
26
55.845
Iron
|
27
58.933
Cobalt
|
28
58.693
Nickel
|
29
63.546
Copper
|
30
65.38
Zinc
|
31
69.723
Gallium
|
32
72.630
Germanium
|
33
74.922
Arsenic
|
34
78.971
Selenium
|
35
79.904
Bromine
|
36
83.798
Krypton
|
|
| 5 |
37
85.468
Rubidium
|
38
87.62
Strontium
|
39
88.906
Yttrium
|
40
91.224
Zirconium
|
41
92.906
Niobium
|
42
95.95
Molybdenum
|
43
Tc ☢
96.906
Technetium
|
44
101.07
Ruthenium
|
45
102.91
Rhodium
|
46
106.42
Palladium
|
47
107.87
Silver
|
48
112.41
Cadmium
|
49
114.82
Indium
|
50
118.71
Tin
|
51
121.76
Antimony
|
52
127.60
Tellurium
|
53
126.90
Iodine
|
54
131.29
Xenon
|
|
| 6 |
55
132.91
Caesium
|
56
137.33
Barium
|
* |
71
174.97
Lutetium
|
72
178.49
Hafnium
|
73
180.95
Tantalum
|
74
183.84
Tungsten
|
75
186.21
Rhenium
|
76
190.23
Osmium
|
77
192.22
Iridium
|
78
195.08
Platinum
|
79
196.97
Gold
|
80
200.59
Mercury
|
81
204.38
Thallium
|
82
207.2
Lead
|
83
208.98
Bismuth
|
84
Po ☢
208.98
Polonium
|
85
At ☢
209.99
Astatine
|
86
Rn ☢
222.02
Radon
|
| 7 |
87
Fr ☢
223.02
Francium
|
88
Ra ☢
226.03
Radium
|
** |
103
Lr ☢
262.11
Lawrencium
|
104
Rf ☢
267.12
Rutherfordium
|
105
Db ☢
270.13
Dubnium
|
106
Sg ☢
269.13
Seaborgium
|
107
Bh ☢
270.13
Bohrium
|
108
Hs ☢
269.13
Hassium
|
109
Mt ☢
278.16
Meitnerium
|
110
Ds ☢
281.17
Darmstadtium
|
111
Rg ☢
281.17
Roentgenium
|
112
Cn ☢
285.18
Copernicium
|
113
Nh ☢
286.18
Nihonium
|
114
Fl ☢
289.19
Flerovium
|
115
Mc ☢
289.20
Moscovium
|
116
Lv ☢
293.20
Livermorium
|
117
Ts ☢
293.21
Tennessine
|
118
Og ☢
294.21
Oganesson
|
| *Lanthanoids | * |
57
138.91
Lanthanum
|
58
140.12
Cerium
|
59
140.91
Praseodymium
|
60
144.24
Neodymium
|
61
Pm ☢
144.91
Promethium
|
62
150.36
Samarium
|
63
151.96
Europium
|
64
157.25
Gadolinium
|
65
158.93
Terbium
|
66
162.50
Dysprosium
|
67
164.93
Holmium
|
68
167.26
Erbium
|
69
168.93
Thulium
|
70
173.05
Ytterbium
|
||||
| **Actinoids | ** |
89
Ac ☢
227.03
Actinium
|
90
Th ☢
232.04
Thorium
|
91
Pa ☢
231.04
Protactinium
|
92
U ☢
238.03
Uranium
|
93
Np ☢
237.05
Neptunium
|
94
Pu ☢
244.06
Plutonium
|
95
Am ☢
243.06
Americium
|
96
Cm ☢
247.07
Curium
|
97
Bk ☢
247.07
Berkelium
|
98
Cf ☢
251.08
Californium
|
99
Es ☢
252.08
Einsteinium
|
100
Fm ☢
257.10
Fermium
|
101
Md ☢
258.10
Mendelevium
|
102
No ☢
259.10
Nobelium
|
||||
The standard form of the periodic table shown here includes periods (shown horizontally) and groups (shown vertically). The properties of elements in groups are similar in some respects to each other.
There is no one single or best structure for the periodic table but by whatever consensus there is, the form used here is very useful and the most common. The periodic table is a masterpiece of organised chemical information and the evolution of chemistry's periodic table into the current form is an astonishing achievement.