Bismuth - 83Bi: the essentials
- Name: bismuth
- Symbol: Bi
- Atomic number: 83
- Relative atomic mass (Ar): 208.98040 (1)
- Standard state: solid at 298 K
- Appearance: lustrous reddish white
- Classification: Metallic
- Group in periodic table: 15
- Group name: Pnictogen
- Period in periodic table: 6
- Block in periodic table: p
- Shell structure: 2.8.18.32.18.5
- CAS Registry: 7440-69-9
Bismuth atoms have 83 electrons and the shell structure is 2.8.18.32.18.5. The ground state electronic configuration of neutral bismuth is [Xe].4f14.5d10.6s2.6p3 and the term symbol of bismuth is 4S3/2.
Bismuth: description
Bismuth is a white, crystalline, brittle metal with a pinkish tinge. Bismuth is the most diamagnetic of all metals, and the thermal conductivity is lower than any metal, except mercury. It has a high electrical resistance, and has the highest Hall effect of any metal (that is, the greatest increase in electrical resistance when placed in a magnetic field).


Cartoon by Nick D Kim ([Science and Ink], used by permission).
Bismuth: physical properties
Density of solid: 9780 kg m-3
Molar volume: 21.31 cm3
Thermal conductivity: 8 W m‑1 K‑1
Bismuth: heat properties
Melting point: 544.4 [271.3 °C (520.3 °F)] K
Boiling point: 1837 [1564 °C (2847 °F)] K
Enthalpy of fusion: 20.5 kJ mol-1
Bismuth: atom sizes
Atomic radius (empirical): 160 pm
Molecular single bond covalent radius: 151 (coordination number 3) ppm
van der Waals radius: [ 280 ] ppm
Bismuth: electronegativities
Pauling electronegativity: 2.02 (Pauling units)
Allred Rochow electronegativity: 1.67 (Pauling units)
Mulliken-Jaffe electronegativity: 2.15 (20% s orbital)
Bismuth: orbital properties
First ionisation energy: 702.95 kJ mol‑1
Second ionisation energy: 1611.6 kJ mol‑1
Third ionisation energy: 2466.5 kJ mol‑1
Bismuth: abundances
Universe: 0.7 ppb by weight
Crustal rocks: 25 ppb by weight
Human: (no data) ppb by weight
Bismuth: crystal structure
Bismuth: biological data
Human abundance by weight: (no data) ppb by weight
Bismuth has no biological role. However it has been used for some time as a medicine (tripotassium dicitratobismuthate) for treatment of stomach upsets. In combination with antibiotics it is now used for treatment of some stomach ulcers. It is also to be found in haemorrhoid creams such as Anusol cream and Hemocaneas as bismuth oxide and in Anusol ointment as bismuth subgallate.
Bismuth: uses
Bismuth: reactions
Reactions of bismuth as the element with air, water, halogens, acids, and bases where known.
Bismuth: binary compounds
Binary compounds with halogens (known as halides), oxygen (known as oxides), hydrogen (known as hydrides), and other compounds of bismuth where known.
Bismuth: compound properties
Bond strengths; lattice energies of bismuth halides, hydrides, oxides (where known); and reduction potentials where known.
Bismuth: history
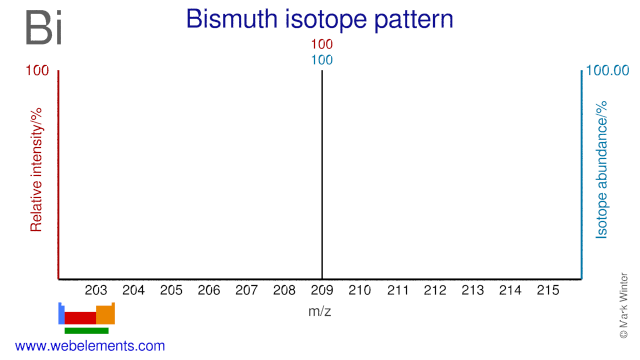
Bismuth was discovered by known since ancient times in unknown at not known. Origin of name: from the German word "bisemutum".Bismuth: isotopes
Bismuth: isolation
Isolation: it is not normally necessary to make bismuth in the laboratory as it is available commercially. Bismuth is found in nature largely as bismite (Bi2O3), bismuthinite (Bi2S3), and bismutite [(BiO)2CO3]. However it is generally made as a byproduct of copper, lead,tin, silver, gold, and zinc plants. The final step involves a reduction of the oxide by charcoal.
