Lithium - 3Li: the essentials
- Name: lithium
- Symbol: Li
- Atomic number: 3
- Relative atomic mass (Ar): 6.94 range: [6.938, 6.997] m [see notes g m r]
- Standard state: solid at 298 K
- Appearance: silvery white/grey
- Classification: Metallic
- Group in periodic table: 1
- Group name: Alkali metal
- Period in periodic table: 2
- Block in periodic table: s
- Shell structure: 2.1
- CAS Registry: 7439-93-2
Lithium atoms have 3 electrons and the shell structure is 2.1. The ground state electronic configuration of neutral lithium is [He].2s1 and the term symbol of lithium is 2S1/2.
Lithium: description
Lithium is a Group 1 (IA) element containing just a single valence electron (1s22s1). Group 1 elements are called "alkali metals". Lithium is a solid only about half as dense as water and lithium metal is the least dense metal. A freshly cut chunk of lithium is silvery, but tarnishes in a minute or so in air to give a grey surface. Its chemistry is dominated by its tendency to lose an electron to form Li+. It is the first element within the second period.
Lithium is mixed (alloyed) with aluminium and magnesium for light-weight alloys, and is also used in batteries, some greases, some glasses, and in medicine.
Lithium does not occur as the free metal in nature because of its high reactivity. Deposits are known all aroun the world. It is a minor component of nearly all igneous rocks and is a component of many natural brines.
Lithium: physical properties
Density of solid: 535 kg m-3
Molar volume: 13.02 cm3
Thermal conductivity: 85 W m‑1 K‑1
Lithium: heat properties
Melting point: 453.69 [180.54 °C (356.97 °F)] K
Boiling point: 1615 [1342 °C (2448 °F)] K
Enthalpy of fusion: 20.5 kJ mol-1
Lithium: atom sizes
Atomic radius (empirical): 145 pm
Molecular single bond covalent radius: 133 (coordination number 1) ppm
van der Waals radius: 212 ppm
Lithium: electronegativities
Pauling electronegativity: 0.98 (Pauling units)
Allred Rochow electronegativity: 0.97 (Pauling units)
Mulliken-Jaffe electronegativity: 0.97 (s orbital)
Lithium: orbital properties
First ionisation energy: 520.22 kJ mol‑1
Second ionisation energy: 7298.16 kJ mol‑1
Third ionisation energy: 11815.05 kJ mol‑1
Lithium: abundances
Universe: 6 ppb by weight
Crustal rocks: 17000 ppb by weight
Human: 30 ppb by weight
Lithium: crystal structure

Lithium: biological data
Human abundance by weight: 30 ppb by weight
Lithium seems to have no biological role, but does have an effect on the body if swallowed. Sometimes, lithium-based compounds such as lithium carbonate (Li2CO3) are used as drugs to treat manic-depressive disorders. The dose is around 0.5 g - 2 g daily.
Lithium: uses
Lithium: reactions
Reactions of lithium as the element with air, water, halogens, acids, and bases where known.
Lithium: binary compounds
Binary compounds with halogens (known as halides), oxygen (known as oxides), hydrogen (known as hydrides), and other compounds of lithium where known.
Lithium: compound properties
Bond strengths; lattice energies of lithium halides, hydrides, oxides (where known); and reduction potentials where known.
Lithium: history
Lithium was discovered by Johan August Arfvedson in 1817 at Stockholm, Sweden. Origin of name: from the Greek word "lithos" meaning "stone", apparently because it was discovered from a mineral source whereas the other two common Group 1 elements, sodium and potassium, were discovered from plant sources..Lithium: isotopes
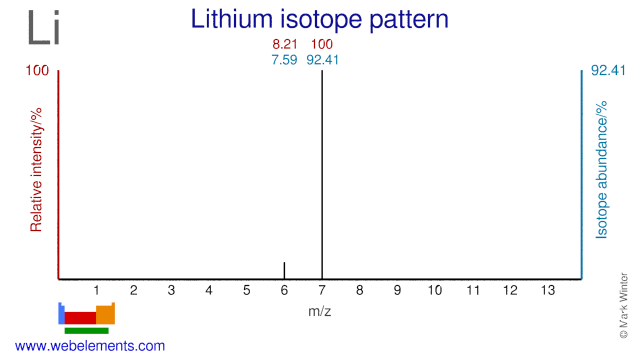
Li-7 is used to control the pH level of the coolant in the primary water circuit of pressurized water reactors. Li-7 is also used for the production of the medical research radioisotope Be-7. Li-6 is used in thermonuclear weapons and the export and use of Li-6 is therefore strictly controlled. Li-6 can also be used for the production of the radioisotope H-3, which is used in biochemistry research.
Lithium: isolation
Isolation: lithium would not normally be made in the laboratory as it is so readily available commercially. All syntheses require an electrolytic step as it is so difficult to add an electron to the poorly electronegative lithium ion Li+.
The ore spodumene, LiAl(SiO3)2, is the most important commercial ore containing lithium. The α form is first converted into the softer β form by heating to around 1100°C. This is mixed carefully with hot sulphuric acid and extracted into water to form lithium sulphate, Li2SO4, solution. The sulphate is washed with sodium carbonate, Na2CO3, to form a precipitate of the relatively insoluble lithium carbonate, Li2CO3.
Li2SO4 + Na2CO3 → Na2SO4 + Li2CO3 (solid)
Reaction of lithium carbonate with HCl then provides lithium chloride, LiCl.
Li2CO3 + 2HCl → 2LiCl + CO2 +H2O
Lithium chloride has a high melting point (> 600°C) meaning that it sould be expensive to melt it in order to carry out the electrolysis. However a mixture of LiCl (55%) and KCl (45%) melts at about 430°C and so much less energy and so expense is required for the electrolysis.
cathode: Li+(l) + e- → Li (l)
anode: Cl-(l) → 1/2Cl2 (g) + e-
