Copper - 29Cu: the essentials
- Name: copper
- Symbol: Cu
- Atomic number: 29
- Relative atomic mass (Ar): 63.546 (3) r [see note r]
- Standard state: solid at 298 K
- Appearance: copper, metallic
- Classification: Metallic
- Group in periodic table: 11
- Group name: Coinage metal
- Period in periodic table: 4
- Block in periodic table: d
- Shell structure: 2.8.18.1
- CAS Registry: 7440-50-8
Copper atoms have 29 electrons and the shell structure is 2.8.18.1. The ground state electronic configuration of neutral copper is [Ar].3d10.4s1 and the term symbol of copper is 2S1/2.
Copper: description
Copper appears in the periodic table at the top of Group 11 above silver and gold. Collectively these are sometimes referred to as the coinage metals, although in modern times these metals are used less frequently than in the past.
Copper is one of the most important metals. Copper is reddish with a bright metallic lustre. It is malleable, ductile, and a good conductor of heat and electricity (second only to silver in electrical conductivity). Its alloys, brass and bronze, are very important. Monel and gun metals also contain copper. Apparently the reason that policemen in the USA are nicknamed "cops" or "coppers" is to do with their uniforms which used to have copper buttons.
Copper metal is not particularly reactive and indeed is found as the free metal ("native") on occasion. Copper sulfate is [Cu(SO4)(OH2)4].H2O in the crystalline state; it is particularly well known and was often called blue vitriol. The oxidation number in most copper compounds is Cu(II), old name cupric, but there are many important Cu(I) compounds (cuprous) as well.

Copper foil.

Copper (I) chloride salts imparts a blue colour to flames. The picture above shows the colour arising from adding cuprous chloride (CuCl) to a burning mixture of potassium chlorate and sucrose. This flame is relatively cool. Hotter flames burn green bacause of emission from copper atoms (only to be demonstrated by a professionally qualified chemist).
Copper: physical properties
Density of solid: 8920 kg m-3
Molar volume: 7.11 cm3
Thermal conductivity: 400 W m‑1 K‑1
Copper: heat properties
Melting point: 1357.77 [1084.62 °C (1984.32 °F)] K
Boiling point: 3200 [2927 °C (5301 °F)] K
Enthalpy of fusion: 20.5 kJ mol-1
Copper: atom sizes
Atomic radius (empirical): 135 pm
Molecular single bond covalent radius: 112 (coordination number 2) ppm
van der Waals radius: 238 ppm
Copper: electronegativities
Pauling electronegativity: 1.90 (Pauling units)
Allred Rochow electronegativity: 1.75 (Pauling units)
Mulliken-Jaffe electronegativity: 1.49 (s orbital)
Copper: orbital properties
First ionisation energy: 745.48 kJ mol‑1
Second ionisation energy: 1957.92 kJ mol‑1
Third ionisation energy: 3554.6 kJ mol‑1
Copper: abundances
Universe: 60 ppb by weight
Crustal rocks: 68000 ppb by weight
Human: 1000 ppb by weight
Copper: crystal structure

Copper: biological data
Human abundance by weight: 1000 ppb by weight
Copper is essential for all life, but only in small quantities. It is the key component of redox enzymes and of haemocyanin.
Copper: uses
Copper: reactions
Reactions of copper as the element with air, water, halogens, acids, and bases where known.
Copper: binary compounds
Binary compounds with halogens (known as halides), oxygen (known as oxides), hydrogen (known as hydrides), and other compounds of copper where known.
Copper: compound properties
Bond strengths; lattice energies of copper halides, hydrides, oxides (where known); and reduction potentials where known.
Copper: history
Copper was discovered by known since ancient times in unknown at not known. Origin of name: from the Latin word "cuprum" meaning the island of "Cyprus".Copper: isotopes
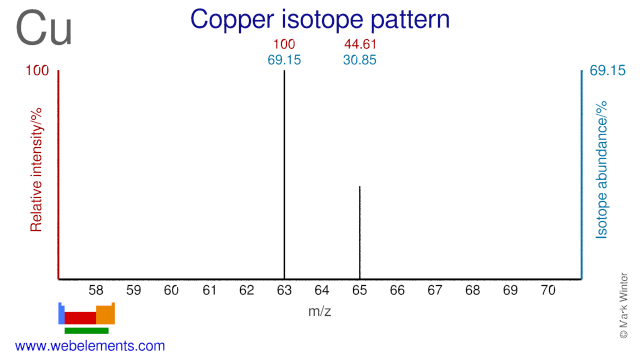
Both isotopes of Copper, Cu-63 and Cu-65, are used to study copper metabolism and gastrointestinal diseases. Cu-63 is used for production of medical radioisotope Zn-62 and can also be used for the production of Cu-64 which is used in cancer diagnosis and treatment. Cu-65 has also been proposed as a precursor for the (cyclotron) production of Cu-64.
Copper: isolation
Isolation: copper metal is readily available commercially so it is not normally necesary to make it in the laboratory. Most copper production is based upon sulphide ores containing little copper but quite a lot of iron. New cleaner technologies are now important but older processes present major environmental problems. Complex procedures are used initially to form a form of copper sulphide appropriate for final reduction via a copper(I) oxide. The resulting crude copper is purified using an electrolytic procedure involving plating onto pure copper cathodes.
2Cu2S + 3O2 → 2Cu2O + 2SO2
2Cu2O + Cu2S → 6Cu + SO2
Notably, the purification step leaves an "anode slime" which contains useful amounts of silver and gold.
